Title of paper under discussion
Early neuronal responses in right limbic structures mediate harmony incongruity processing in musical experts
Authors
Clara E. James, Juliane Britz, Patrik Vuilleumier, Claude-Alain Hauert, Christoph M. Michel
Journal
NeuroImage, Volume 42, Issue 4, 1 October 2008, Pages 1597-1608
Overview
Reckoning that interrupted cadences “should evoke a profound and rapid reaction” in a listening musician’s brain, the authors commissioned a set of short piano excerpts each of which either ended either with a perfect or an interrupted cadence (referred to in the paper as ‘closure congruity’ or ‘closure incongruity’). The works were played to two groups, expert pianists and laymen, whose brain activity was being monitored using electroencephalography (EEG). It turned out that the musicians alone demonstrated a strong negative ‘Event Related Potential’ (ERP) skewed towards the right hemisphere, about 200ms after the interrupted cadence (the ‘event’), with further analysis suggesting this response was based in right limbic (emotions and memory) regions: hippocampus, amygdala and insula.
Introduction
Plenty of research has detailed changes in the auditory and motor areas of the brain in response to musical training, but what of cognitive and emotional brain regions – do these areas also function differently in musicians, displaying a different pattern of brain activity whilst listening to music compared with the same brain areas in musical ‘laymen’?
As a research window into cognitive and emotional music processing the authors chose to compare brain activity in musical experts and laymen immediately after listening to an interrupted cadence, hypothesising that this ‘subtle transgression of musical grammar’ might be ‘apprehended by musical experts’ but not by laymen.
In order to be sure of evoking the most ‘natural and complete responses’ possible the researchers paid a distinguished composer (Nikolaas Ravenstijn) to write ‘a series of expressive polyphonic piano pieces’ that offered a ‘rich and expressive musical context’, each of which had two alternate endings: a perfect or an interrupted cadence. The two groups – musical experts and laymen – listened to these pieces as their brain activity was monitored by EEG electrodes, and were asked to choose whether the ending was ‘satisfactory or unsatisfactory’. If their hypothesis was correct then the musicians should be better at spotting the interrupted endings – and should display distinctive patterns of brain activity in response to them.
Previous research on ‘syntactically irregular chords’ had been shown to evoke in listeners (musicians and non-musicians alike) a negative electrical potential in the front right area of the scalp very soon after the event, called an ‘early right-anterior negativity’ or ERAN. The authors hoped that a similar brain reaction might be observed here, and perhaps just in musicians due to the subtlety of the ‘transgression’.
Method
26 male volunteers (all male because neurophysiological responses to music are known to be sex sensitive) were recruited – 13 expert pianists (from Swiss and French conservatoires) and 13 musical laymen.
30 excerpts of solo piano music were expressly composed, performed and recorded (not synthesised!) for the experiment, each with two alternative final cadences, one perfect and one interrupted (thus making 60 excerpts). Each participant listened to all the excerpts (in random order), each one 3 times, so that the playlist was 180 excerpts long. As the experiment progressed listeners were asked to indicate whether each final cadence sounded ‘satisfactory’ or ‘unsatisfactory’.
(I write ‘interrupted cadence’ for the sake of brevity. Strictly speaking only two thirds of the cadences were textbook ‘interrupted’, meaning they fell on the submediant. The other third fell on the subdominant, not strictly ‘interrupted’ but certainly ‘unsatisfactory’).
Here are two examples of the musical excerpts, each with its alternative perfect cadence (marked ‘R’ for regular) and interrupted cadence (‘I’ for incongruous):


All the while the listeners’ brain activity was being recorded, more specifically the electrical potential (in microvolts) at each of 128 superficial electrodes evenly distributed around the scalp and measured over time (in milliseconds) from the moment the final cadence was played to half a second later. Such readings are called ‘Event-Related Potentials’ (ERPs), the ‘event’ in question here being the final cadence.
Analysis of the EEG data was carried out in three stages: 1) A ‘first step’ investigation – using data from just 5 electrodes and looking only at a time-window of 200-260 ms after the cadence – comparing EEG readings in musicians and laymen after perfect and interrupted cadences, especially looking for evidence of an ‘early right-anterior negativity’ (ERAN) after an interrupted cadence 2) An analysis called ‘microstate segmentation’ whereby the progress over time of the topographical map of the scalp’s electric field is presented in a series of discrete snapshots (or ‘microstates’) rather than as constantly changing movie and 3) an attempt to locate the sources in the brain responsible for any differences in the EEG readings between musicians and laymen.
Results
When it came to identifying the final cadences musical laymen scored only just above chance (66% correct answers) in their ability to identify an interrupted (‘unsatisfactory’) cadence on hearing it, whereas the musicians scored 96%. Perfect (‘satisfactory’) cadences proved easier to recognise – 81% (laymen) and 96% again (musicians).
Regarding analysis of EEG data in the three stages listed above:
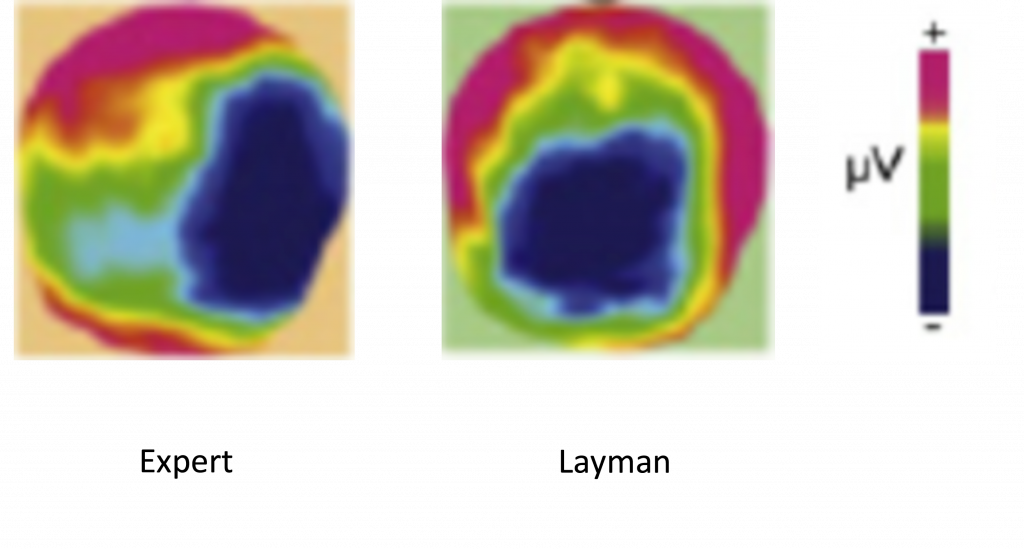
1) The ‘first step’ five-electrode analysis revealed what the researchers had been hoping for: signs of an early right-sided negative component in relation to the interrupted cadence… but only in musicians. The graphs below show the EEG readings at each of the five electrodes and compare readings (in microvolts) obtained in the milliseconds following a perfect cadence (top) and after an interrupted cadence (below). Within each graph is a comparison between musical experts (in red) and laymen (in black).
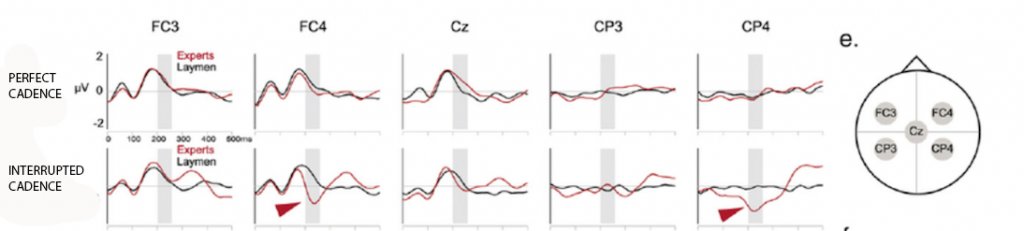
The graphs demonstrate that there is little difference between musicians and laymen when it comes to EEG readings following a perfect cadence. However about 200ms after an interrupted cadence the two groups show a marked difference: there are two electrodes especially – FC4 and CP4, both on the right side of the head – where there is a marked negativity in the musicians’ readings at this time, marked with red arrows on the graph.
Comparing top and bottom graphs it’s also discernible that there was little difference between the EEG readings following a perfect cadence and an interrupted cadence in laymen; but that there was a strong difference between the two sets of readings in musicians, due to the strong right-sided negative component after the interrupted cadence. This next graphic compares this ‘Interrupted cadence minus regular (perfect) cadence’ EEG difference in musicians (‘experts’) and laymen, with the strong early right sided negativity showing up as a large dark blue patch in experts:

2) On hearing the final cadence, be it perfect or interrupted, the progress (for the next 500 milliseconds) of readings from each of the 128 electrodes was recorded, and those readings were then collated and turned into continuously changing topographical maps representing EEG readings across the scalp. These ‘map movies’ were segmented into snapshots called ‘microstates’ – such segmentation actually happens naturally, as the “topography of the electric field at the scalp does not very randomly as a function of time, but rather remains in a stable configuration for brief time periods”.
Here are the ERP (event-related potential) and microstate data presented over those 500ms, both of experts (top) and of laymen (bottom), after a perfect cadence:
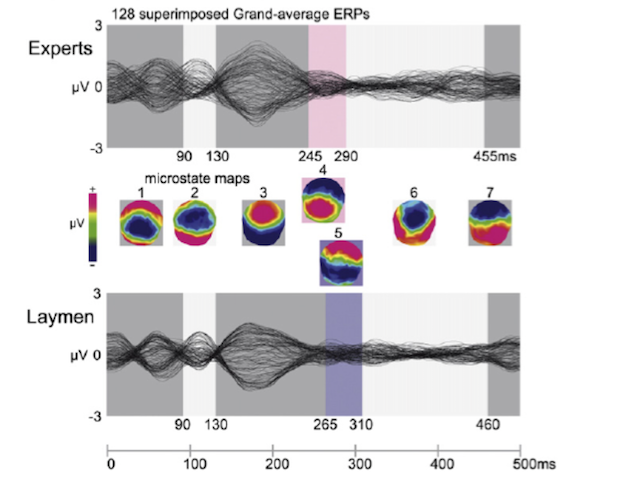
It’s clear on inspection that the two groups share similar data, and that they have nearly all the resultant microstate maps (1,2,3,6,7) in common – only state 4 is peculiar to musicians and state 5 to laymen.
Now, here is the same comparison but after an interrupted cadence:
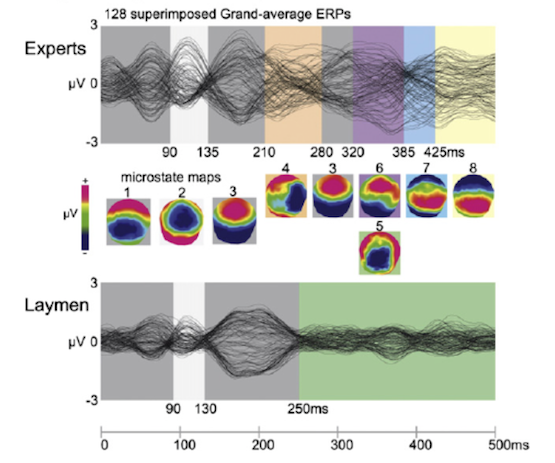
Notice that up until about 200 milliseconds after the event (the interrupted cadence) all listeners share the same series of microstates (1,2,3) but from 200 ms onwards the two groups have wildly different patterns of brain activity: musicians cascade along five different states (4,3,6,7,8) while laymen remain steady in one state (5). See also that state 4 demonstrates the ‘right negativity’ reminiscent of the ERAN (early right-anterior negativity) seen in previous research.
The later microstates – 3,6,7 and 8 – show musicians developing positive (red) ERPs starting at the front of the brain and moving towards the back over time. This is in contrast to laymen whose single microstate (5) is characterised by “posterior negativity” (coloured blue).
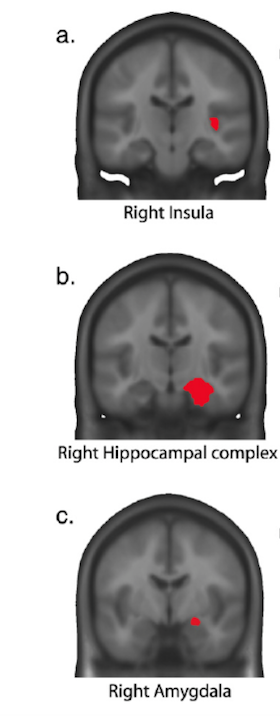
3) In their quest to find out which specific brain structures were responsible for these differing EEG readings the researchers carried out a complex statistical analysis looking at fifty ‘regions of interest’ in the brain and identified three that were “differentially activated” after interrupted cadences in musicians as compared with laymen – the right insula, right hippocampus and right amygdala, all parts of the limbic system and all shown in the diagram below:
Discussion
The authors identify the “key finding” of this study as “a selective rapid neuronal response (starting early as c200ms) to subtle chord violations specifically for trained pianists, but not for musical laymen.”
They go on to describe how “this rapid response manifested as a right lateralised negative ERP component” and that “right temporal-limbic areas, encompassing hippocampal complex and amygdala, and right insula, brain areas critically associated with cognitive, memory and emotive processing, contributed to the generation of this early negative ERP.”
In contrast, laymen merely showed a “symmetric posterior negativity” (see the blue patch at the back of the scalp in micro states 3 and 5) in response to the interrupted cadence.
When comparing this “key finding” with the ERAN described by previous researchers, the authors concluded that although it had similarities (being early, negative, and right-sided) it was distinguished by being strongest in the temporal region, completely right lateralised, seemingly due to medial-temporal brain structures (insula, hippocampus and amygdala), and – crucially – only visible in musical experts.
The authors hypothesise that the involvement of musically developed limbic structures – insula, hippocampus and amygdala – may subserve “enhanced higher-order pitch and tonality processing together with a highly specific auditory memory for musical syntax”, in addition to emotional activity due to “frustration or disliking” from “subtle expectancy violation”. They make clear that this is a quick response, perhaps allowing for “rapid adaptation and error correction” in an expert musician.
The right-sided lateralisation of the response tallies with previous research describing the right hemisphere’s dominance in auditory and medial-temporal areas for tonality processing (with the left hemisphere more involved in speech).
It’s suggested that the later microstates in the musician – 3,6,7 and 8, showing an area of positivity moving from front to back over time – reveal activation in motor parts of the brain (reflecting automatic motor involvement), the anterior cingulate cortex (perhaps reflecting error detection and decision making), orbitofrontal cortex (emotion) and visual cortex (reflecting visual imagery during music processing).
In conclusion, the paper describes how the limbic system of musicians – rather than the auditory system – seems to carrying out the early cognitive and emotive analysis of chord function, demonstrating how it’s not only auditory and motor areas of the brain that show ‘functional plasticity’ in response to musical training, but also the limbic structures.
Coda
A Musical Joke – W A Mozart (1756-91)
Liszt Ferenc Chamber Orchestra
Please can you also do a study of what happens to the brain, when subjected to non-stop beats, or direct me to it, if already done? We didn’t have to be surrounded by ‘music’ driven by beats when I was started to play the cello!
Hi Celia, thank you for the comment (now revealed after dealing with spam, sorry for the delay!) – take a look at this fascinating paper on the basal ganglia for more information on how we internally generate a beat (compared with relying on externally presented ‘non-stop beats’): http://www.jessicagrahn.com/uploads/6/0/8/5/6085172/34.pdf
I noticed that the interrupted cadence, at least the one shown here, is problematical, as it also has problematical voice leading (parallel 5ths) which is immediately audible – so this could add to the auditory ‘discomfort’ that musicians, rather than laymen, feel… Seems the composer did not take this into account, or the researchers…
Hi Mark, Ouch – good spot! Every lab needs a Riemenschneider.. thanks for comment, Adrian